As discussed in this author’s last 2 posts, the common cold (rhinovirus) was covered as well as its negative affects upon children, adults, and cellular function. Quercetin and zinc were explored as potential interventions to help limit viral entry into cells, replication, and inflammatory cascades. In the following sections, this author will explore vitamin C (VC) as another means of supporting the immune system, and overall cellular function, during the common cold.
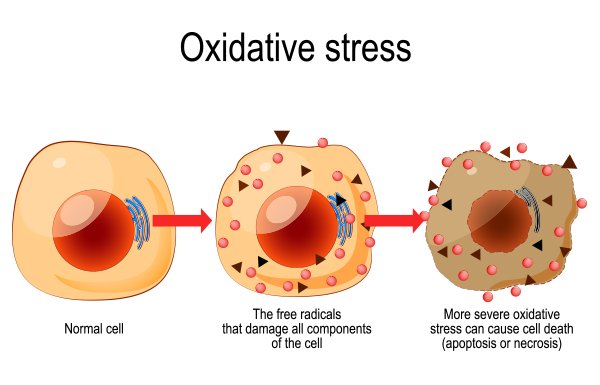
VC, also known as ascorbic acid, was isolated and discovered in 1928 and widely known for its ability to correct scurvy; a condition induced by VC insufficiency.1 VC is found primarily in fruits and vegetables to include strawberries, lemons, grapefruit, oranges, papaya, cantaloupe, kale, green pepper, brussel sprouts, asparagus, broccoli, and cauliflower.1(304) VC is known to play complex roles within the body and functions as a cosubstrate for enzyme activity and as an antioxidant. Please see the link below for more information on antioxidants and oxidative stress:
Click: Antioxidants
VC has been shown to induce positive effects upon several components of both the innate and adaptive immune system. Furthermore, said vitamin not only supports the immune system against rhinovirus (common cold) but might also help manage coronaviruses.2 It should be noted that regular consumption of aforementioned foods will supply levels of VC deemed adequate (i.e., 100-200 mg/day) to maintain normal immune function. However, research has suggested that established infections requires significantly higher (gram) doses of vitamin C to optimize and support immune function.3
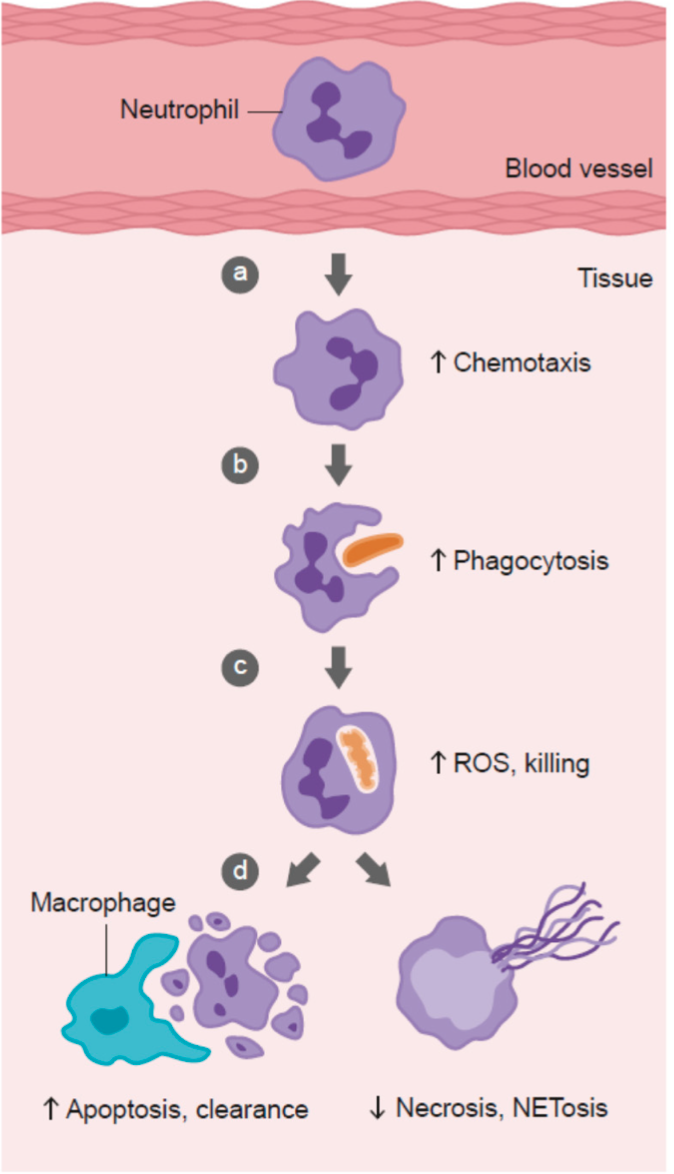
Neutrophils are white blood cells serving as a first line of defense (innate immunity), which consumes, digests, and kills pathogens using antimicrobial peptides, proteins, and reactive oxygen species.3(7) Macrophages, another form of white blood cell and also a part of innate immunity, consumes/kills pathogens, removes spent neutrophils (discussed later), and sends signals to other cells to fight infections. VC has been shown to enhance chemotaxis (movement) of neutrophils towards a pathogen; a key step in neutralizing a virus.3(4) VC has also been shown to improve phagocytosis (envelopment and entrapment of the virus) and apoptosis (programmed cell death) of spent neutrophils.3(4)
Apoptosis of neutrophils, after they neutralize a pathogen, may seem counterintuitive. However, such is relevant as spent/used neutrophils who remain “alive” can induce further cell damage (by releasing their anti-pathogenic arsenal) and inflammation if left unchecked.3(4) VC promotes planned death of the neutrophil and allows safe removal of the same via macrophages. Furthermore, VC not only improves chemotaxis and apoptosis; VC is thought to protect the neutrophil from oxidation while it showers its target pathogen with reactive oxygen species; a process which optimizes neutrophil function during mitigation of infection.3(4) It should be noted, however, that gram doses (i.e., up to 4 grams/day) of VC are required for enhanced chemotaxis/expedited recovery from cold symptoms.3(6),7
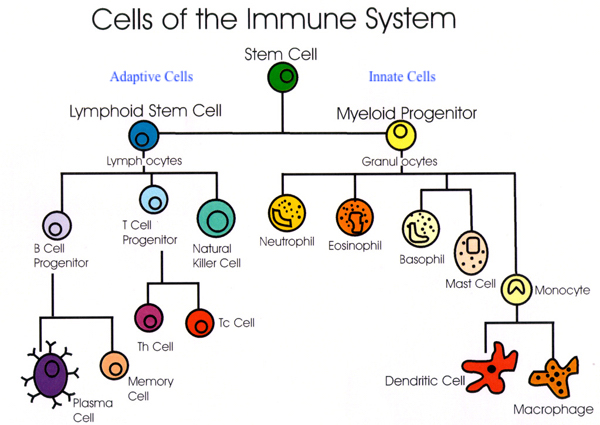
Lymphocytes are white blood cells of the adaptive immune system which include B-cells, T-cells, and natural killer cells (see diagram above). Interestingly, B and T cells both accumulate VC at high levels. In-vitro (cell culture) studies have indicated that VC helps in the proliferation of lymphocytes; such is relevant because lymphocytes are cells, which produce antibodies.3(9) Said studies have also indicated that VC has a central role in the development/maturation of T-cells (helper T-cells and cytotoxic T-cells) and natural killer cells.3(9) The following sections will consider said lymphocytes in greater detail.
Helper T-cells are primarily used for sending signals via releasing interferon-gamma, interleukin-2, and tumor necrosis factor; messengers, which attract other immune system cells to an infected site (macrophages, B-cells, cytotoxic T-cells).4Cytotoxic T-cells bind to infected cells and release cytotoxins, which signals apoptosis (death) of the cell.5 However, cytotoxic T-cells require stimulation and guidance to infected cells by antigen presenting cells. Natural killer cells, however, do not require antigen presenting cells and are thusly able to more quickly than cytotoxic T-cells. Natural killer cells identify virally infected cells, release cytokines (messengers) to include interferon gamma and tumor necrosis factor alpha, which activate macrophage and dendritic cells to facilitate destruction and removal the infected cell.6
In conclusion, optimal levels of VC help support both the innate and adaptive immune system. Specifically, VC has been shown to improve chemotaxis (movement of neutrophils towards a pathogen), phagocytosis (engulfment/entrapment of a pathogen via neutrophils and macrophages), reactive oxygen species generation (to disable the pathogen), protection (thought to protect the neutrophil membrane), growth and spread of B and T-cells, and ultimate destruction of the pathogen. As such, when an individual is exhibiting symptoms of the cold, supplemental VC may help support the immune system in resolving said viral infection.
References
1. Gropper SS, Smith JL, Carr, TP. Advanced Nutrition and Human Metabolism. 7th ed. Boston, MA: Cengage Learning; 2018.
2. Hoang BX, Shaw G, Fang W, et al. Possible application of high-dose vitamin C in the prevention and therapy of corona virus infection. J Glob Antimicrob Resist. 2020:256-262. doi:10.1016/j.jgar.2020.09.025.
3. Carr AC, Maggini S. Vitamin C and immune function. Nutrients. 2017;9(11):1-25. doi: 10.3390/nu9111211.
4. Alberts B, Johnson A, Lewis J, et al. Molecular Biology of the Cell. 4th ed. New York: Garland Science; 2002. https://www.ncbi.nlm.nih.gov/books/NBK26827/. Accessed September 21, 2021.
5. Janeway CA, Travers P, Walport M, et al. The Immune System in Health and Disease. 5th ed. New York: Garland Science; 2001. https://www.ncbi.nlm.nih.gov/books/NBK27101/. Accessed September 21, 2021.
6. Eissmann P. Natural Killer Cells. British Society for Immunology. Accessed September 21, 2021. https://www.immunology.org/public-information/bitesized-immunology/cells/natural-killer-cells.
7. Ran L, Zhao W, Wang J, et al. Extra dose of vitamin C based on a daily consumption shortens the common cold: A meta-analysis of 9 randomized controlled trials. Biomed Res Int. 2018:1-13. doi:https://doi.org/10.1155/2018/1837634.
-Michael McIsaac