As mentioned in this author’s previous posts, Crohn’s disease (CD) is a condition characterized by chronic and inflammatory episodes, which can affect any section of the digestive tract.1 Thus far, this author has outlined steps to reduce inflammation, improve digestion/absorption, reduce intestinal permeability, and improve repair of the gastrointestinal tract. In the following sections, vitamin D3 (VD3) will be explored as another low-cost and low-risk means of helping manage CD symptoms.
VD3 has a widely understood function and role in maintaining bone health; paradoxically, VD3 insufficiency remains common.2 Such insufficiency tends to increase as populations move towards northern climates, as well as the risk of developing CD.2(295) Though such observations are associative in nature, said trends does encourage one to consider the role of low VD3 as a risk factor for developing CD.
SUMMARY
Vitamin D levels (25OHD) tend to decrease as populations move further north of the equator
CD prevalence also tends to increase as populations move further from the equator
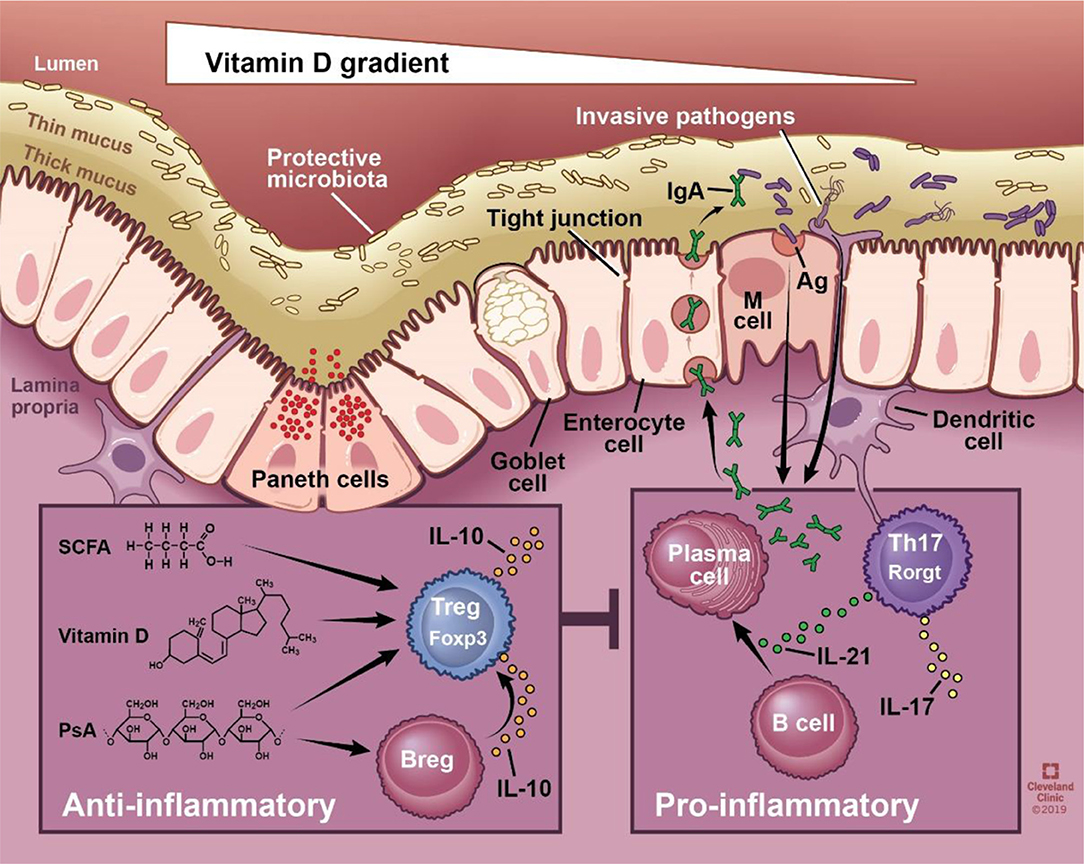
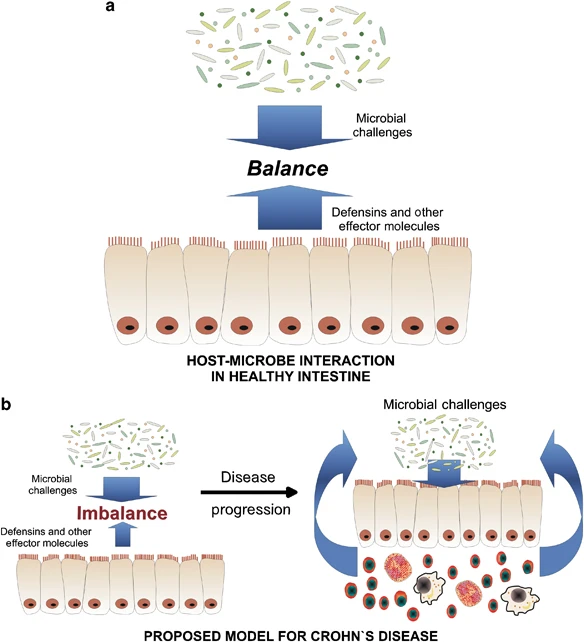
Interestingly, Raftery et al2(295) stated that several studies using experimental models have supported the positive effects of VD3 upon inflammation and inflammatory markers associated with inflammatory bowel disease (IBD); a general term of which CD falls under. Interestingly, little research has explored the effects of VD3 upon intestinal permeability (IP), which is a constituent and aggravator of CD.2(295) Other studies have indicated that VD3, once inside the body (known as 25OHD), has been shown to increase tight junction (TJ) activity; proteins known as zonula occludens and E-cadherin, which maintain intestinal integrity and prevent pathogens and large food molecules from entering the bloodstream.2(295) Not only can VD3 preserve barrier integrity; said vitamin may also indirectly preserve the same through modulation of immune mechanisms.2(295)
SUMMARY
Several studies support the positive role of vitamin D against inflammatory bowel disease
Vitamin D helps maintain optimal intestinal permeability
Vitamin D helps control immune responses
Two antimicrobial peptides known as human cathelicidin (LL37) and beta defensins are components of the innate immune system, which help protect the gut lining from pathogens.2(295) Said peptides are also known as anti-microbial peptides (AMPs) which are expressed directly from the gastrointestinal epithelium; LL37 also promotes wound healing along the intestinal cells (enterocytes) and reduce associated inflammation.2(295) VD3 is thought to influences said AMPs. In vitro (outside of the body) studies have suggested that VD3 induces expression of genes (hCAP18) that encode for LL37, and that LL37 concentrations increase with VD3 supplementation within clinical tuberculosis studies.2(295) Raftery et al2(295) conducted an experiment where VD3 supplementation was used to examine LL37 concentrations and associated IP among individuals with CD.
SUMMARY
Human cathelicidin (LL37) and beta defensins are part of the gut barrier
They stop pathogens from gaining entry into the bloodstream
Also promote wound healing
They reduce inflammation
Vitamin D is suggested to express synthesis of human cathelicidin (LL37) and beta defensins
27 participants with CD were recruited for the researchers’ double-blind placebo-controlled study. Participants for the VD3 group (intervention group) and placebo (control) group were randomized and provided with a 3-month supply of VD3 or a placebo. Baseline 25OHD was measured before the study, and after the study for comparison while the dose of VD3 was 2000 IU/day/person (in the intervention group). IP was measured by the lactulose/mannitol test, sucrose test, and sucralose tests before, and after, the study.2(296) Finally, LL37 was measured at baseline, and after, the study in both groups.2(296) Results indicated that IP increased significantly in the control group over 12 weeks, yet remained stable in the intervention group, though they did not improve.2(300) LL37 concentrations lowered in the control group while they increased within the intervention group. Furthermore, individuals with higher 25OHD concentrations had higher LL37, suggesting 25OHD levels between 100-175 nmol/L may be necessary to exert deeper immune-modulating effects.2(300)
SUMMARY
Vitamin D (2000 IU/day) supplementation increased concentrations of human cathelicidin
Higher concentrations of human cathelicidin were associated with higher blood levels of vitamin D (25OHD)
In conclusion, CD is a condition characterized by chronic and inflammatory episodes, which can negatively affect an individual’s digestive tract function, health, and overall quality of life. In addition to implementing the four R protocol (FRP), use of VD3 in the amount of 2000 IU/day may help stabilize IP; a constituent and aggravator of CD. When combined with the FRP, it is likely that signs and symptoms of CD will be minimized. Ultimately, such an inclusive approach should help support the recovery of individuals living with such an inflammatory bowel disorder.
References
1. Benjamin J, Makharia G, Ahuja V. Glutamine and whey protein improve intestinal permeability and morphology in patients with Crohn’s disease: A randomized controlled trial.Dig Dis Sci. 2012;57:1000-1012. doi: 10.1007/s10620-011-1947-9.
2. Raftery T, Martineau AR, Greiller CL, et al. Effects of vitamin D supplementation on intestinal permeability, cathelicidin and disease markers in Crohn’s disease: Results from a randomised double-blind placebo-controlled study. United European Gastroenterol J. 2015;3(3):294-302. doi: 10.1177/2050640615572176.
-Michael McIsaac